Caracterizarea fizico-chimică ale unor băuturi lactate bazate pe LactoSer
Journal of Environmental Protection and Ecology 19, No 2, 686–693 (2018)
D. Prodana, M. Filipa*, M. Moldovana, I. Perhaițaa, F. Scurtuc, L. Silaghi Dumitrescuaa, A. Dreancab, I. Marcusb
a "Raluca Ripan" Institute for Research in Chemistry, Babes-Bolyai University, 30 Fantanele Street, 400 294 Cluj-Napoca, Romania E-mail: filip_miuta@yahoo.com
b Faculty of Veterinary Medicine, University of Agricultural Sciences and Veterinary Medicine Cluj-Napoca, 3–5 Manastur Street, 400 372 Cluj-Napoca, Romania
c Faculty of Chemistry and Chemical Engineering, Babea-Bolyai University, Arany Janos Street, 400 024 Cluj-Napoca, Romania
Abstract
Aims and background
Milk whey (also called lactoserum) has traditionally been considered a by-product of cheese-making and casein manufacture; more recently, however, the milk whey has been reconsidered as an excellent source of bioactive proteins, lactose, vitamins, minerals and lipids, as opposed to a simple waste material1. Various types of milk whey have been described, with relatively similar chemical compositions but varying widely in terms of preparation procedures in so far as such factors are involved as pretreatment of the cheese milk (heating, centrifugation, cultures used, use of membrane processes, etc.) and the whey handling and pretreatment processes (pasteurisation, pre concentration, recovery of casein fines)2,3. The cheese whey and probiotic cultures use in the preparation of dairy beverages has been extremely attractive by food biotechnology industry4. A new option to add further value to dairy beverages can be supplementation with probiotic bacteria and prebiotic ingredients5 with potential health benefits. The use of whey in the production of dairy beverages could be an alternative for dairy industries6.
In the present study, a commercial formula of milk whey (lactoserum namely Zonar) obtained from cow milk is characterised regarding physicochemical composition. This preparation is intended to use in curative purposes, at nutritional therapies, or as an adjuvant in the treatment of various diseases such as obesity or type II diabetes. Various added ingredients (ginger, cacao powder, honey, colloidal silver solution) have been described in whey for obtaining new dairy beverage with wider use in various diseases. Beverages with bioactive compounds (from extracts of cocoa, Hibiscus flower, ginger) are consumed for their food value and the health benefit for consumers7. Ginger is a potential herbal alternative in the management of digestive disorders, migraines, painful arthritis, and motion sickness8. Cocoa powder contains flavonoids that help prevent systemic inflammation, improves blood flow and helps lower blood pressure. Honey can serve as raw material/substrate for the production of kefir-like beverages with functional and flavouring properties as well as high antioxidant activity, low lactose content and potential probiotic composition9. Also, it has been seen that if silver nanoparticles (AgNps) are directly added into milk the microbial growth decreased10.
The purpose of this study is to investigate the physicochemical characteristics of some new dairy beverages based on lactoserum Zonar and of its three variants with added ingredients such as ginger, honey, cocoa powder and colloidal silver solution, which were recently launched on the Romanian market.
Experimental
Measurement of pH and titratable acidity (TA, Thorner degrees). The pH was determined using a Hanna Instrument pH 211 Microprocessor pH Meter; the overall acidity of samples was determined by an analytical method in according STAS 6353-75, using the following equation:
(1)TA (°T) = (V/10) × 100,
where V is the volume of 0.1 N NaOH solution used for titrationin cm3, and 10 – the sample volume taken in the analysis in cm3.
Determination of water content and total dry matter. A ceramic capsule was dried in an oven at 103 ± 2°C for at least 1 h. After cooling in a desiccator, the capsule was weighed and 10 ml of milk/lactoserum sample were added. The capsule was weighed again, then dried in the oven at 103 ± 2° C for 4–5 h and, after cooling in a desiccator, weighed again. The drying is repeated, until a constant mass was reached. The water content (Water, %) and total solid content (T.S., %) are calculated as follows:
(2)Water (%) = (m – m1)/m2 × 100
(3)T.S. (%) = 100 – Water (%)
where m is the mass in mg of the capsule with the sample prior to drying; m1 – mass in mg of the capsule with the sample after drying; m2 – mass in mg of initial sample added to the capsule.
Determination of sodium chloride content. Aliquots of 25 g of each sample were analysed, after shaking in order to ensure homogeneity. Determination was done by precipitation11 with a AgNO3 solution in the presence of potassium chromate as indicator until the colour turned red-brick. The sodium chloride (NaCl, %) content is calculated using the following equation:
(3)NaCl (%) = 0.00585V/m
where 0.00585 is mg of NaCl corresponding to 1 ml of 0.1 N AgNO3 solution; V – the volume of 0.1 N AgNO3 solution, ml, used for titration; m – mg of lactoserum sample, taken into work.
Determination of lactose content by High-Performance Liquid Chromatography (HPLC)
Lactose was purchased from Aldrich (Milwaukee, USA).
HPLC sample preparation. 1 ml of milk-serum sample was diluted with 4 ml water. To 1 ml of this diluted solution 4 ml of 70% of acetonitrile were added, leading to protein precipitation; the solution was passed through a 0.45 μm filter and injected in the HPLC system. A 20.15 mg ml–1 stock solution of lactose was prepared in ultrapure water. The lactose calibration curve includes 5 concentration points between 20.15 and 1.0075 mg ml–1.
Equipment and method. A HPLC Jasco Chromatograph (Japan) with a refractive index detector was employed. The data were processed with the ChromPass software. Separation was carried out on a CARBOSep column at 70ºC column temperature. The mobile phase was a Milipore ultrapure water; the flow rate was 0.5 ml min–1.
Determination of total proteins by Electrophoresis
For comparison, samples of commercial milk and of whey derived from commercial milk by simple addition of acetic acid were also examined.
Preparation of Bradford reagent. 100 mg Coomassie Brilliant Blue G-250 were dissolved in 50 ml 95% ethanol and 100 ml 85% (w/v) phosphoric acid was added under continuous stirring. The resulting solution was diluted to a final volume of 1 l with final concentration of the reagent: 0.01% (w/v) Coomassie Brilliant Blue G-250, 4.7% (w/v) ethanol, and 8.5% (w/v) phosphoric acid12. Bovine serum albumin was employed for the calibration curve. The absorbance was monitored at 595 nm, using a Perkin Elmer Spectrophotometer. For polyacrylamide gel electrophoresis under denaturing conditions (15% SDS-PAGE), 10 μl of a 20 μg ml–1 final concentration of denatured protein sample were loaded on the gel.
determination of minerals by Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES)
Determination of minerals Ca, K, Mg, Na, P, Cu, Fe, Zn, Pb and Ag from the lactoserum samples was performed using the dry digestion method and an Optima 2100 inductively coupled optical emission spectrometer (ICP-AES).
Sample preparation. Aliquots of 5 g of pre-mixed samples were analysed, in triplicates. The crucibles with sample were placed in a thermostatic oven at 100°C to evaporate the water; after drying, they were placed in a thermostated electric furnace, raising the temperature gradually to a 550°C temperature where it was maintained for 7 h. After the samples were removed from the furnace, they were treated with a mixture of concentrated nitric acid (65% HNO3) and hydrogen peroxide (30% H2O2) in 1: 0.6 ratio of successively added to each sample. After addition of HNO3, the sample crucibles were placed on a sand bath to evaporate gradually. After cooling, H2O2 was added to the samples, and heating was continued on the sand bath until the samples had dried. The operation was repeated two more times.
Results and discussion
The serum phase represents the watery portion of milk, without fat globules and casein micelles. The whey proteins are located in solution in the serum phase of milk along with minor proteins and enzymes, including lacto peroxidase, lacto transferrin, lysozyme, glycoprotein, serum transferrin, and degradation products derived from casein. The whey proteins and caseins are distinguishable from each other by their physical and chemical characteristics such as: the whey proteins present hydrophobic and hydrophilic regions while the casein presents strong hydrophobic regions; the whey proteins are stable in mild acidic environmental while the caseins precipitate in acidic conditions and insoluble at pH 4.6. The whey proteins have water-binding capacity, increases with denaturation of the protein, are soluble at all pH levels; insoluble at pH 5, if they are denatured. They have a low viscosity for native protein and higher if denatured. The gelation occurs at 70°C (158°F) or higher and is influenced by pH and salts13.
Figure 1 shows SDS-PAGE data on Zonar, as compared to bovine milk and to a whey derived there from by a classical procedure. The two dominant proteins are, in line with previous studies on other whey preparations14,15, α-lactalbumin (14 kDa) and β-lactoglobulin (18 kDa). This profile is similar to the one displayed by a reference whey sample prepared in the laboratory by simple chemical treatment, and attributable as "weet whey"15.
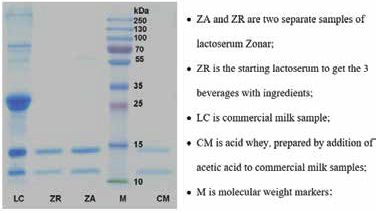
Fig. 1 SDS-PAGE gel of two samples of lactoserum Zonar compared to commercial milk and to whey derived therefrom by simple precipitation with acid
Lactose is the main component of whey, constitutes about 4.4–4.9% of the whey ‘as is’ (almost 75% of the dry matter) depending on the whey type16. Lactose content was obtained by HPLC-RI (refractive index detection) method. The calibration curve (linear range 20.15–1.0075 μg ml–1) has the regression equation Y = 0.0065X + 1.74 and regression coefficient of 0.9989. The limits of detection (LOD) and quantification (LOQ) were 13.7 and 41.1 μg ml–1, respectively. In Fig. 2 are shown the HPLC-RI chromatograms of lactose standard and Zonar sample.
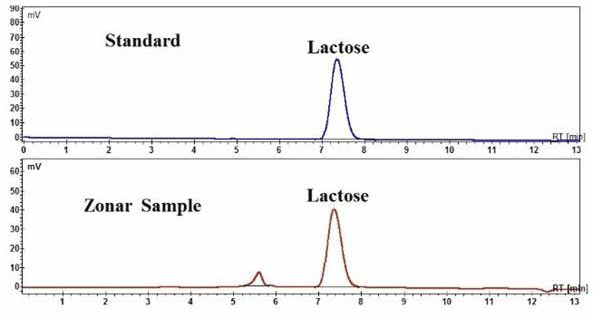
Fig. 2 HPLC-RI chromatograms of lactose standard (RT = 7.24 min) and lactoserum Zonar
Table 1 shows the physicochemical characteristics of the four types of milk serum dairy beverages.
| Characteristics | Z | Z-GE-H | Z-CE-H | Z-CS | M-W17, 18 |
|---|---|---|---|---|---|
| pH | 6.2 | 6.1 | 6.2 | 6.3 | 6.2–6.4 |
| Acidities (ᵒT) | 9.0 | 10.0 | 10.9 | 9.9 | 4 [SH]* |
| Dry matter (%) | 6.3 | 6.1 | 6.1 | 6.2 | 6–6.5 |
| Water (%) | 93.7 | 93.9 | 93.9 | 93.8 | 93–94 |
| NaCl (%) | 0.2 | 0.19 | 0.19 | 0.19 | - |
| Lactose (%) | 4.8 | 4.7 | 4.7 | 4.6 | 4.5–5 |
| Total protein of lactoserum Zonar (μg/ml)** | |||||
| centrifuged 15 min | 538 | ||||
| centrifuged 3–5 min | 781 | ||||
| not centrifuged | 2170 | ||||
Table 1. Physicochemical characteristics of new dairy lactoserum Zonar samples
M-W – milk way content present in literature data; Z – lactoserum Zonar; Z-GE-H – Zonar with ginger extract and honey; Z-CE-H – Zonar with cocoa extract and honey; Z-CS – Zonar with colloidal silver solution; *[SH] = 2.5ᵒT; ** 0.6–0.65% for sweet whey19; centrifugation was performed in order to remove particulate matter (proteins, cells).
In order to achieve a stable formulation starting from the basic properties of the whey and in order to improve them, an attempt was made to reduce the salt content. Also, the mineral substances in the concentrate, especially the calcium salts, increase the flocculation of serum proteins from the concentrate on heating, while the citrate and phosphate anions protect Ca2+,increasing the thermal stability of the proteins to a neutral pH.
The mineral composition differs most with the pH and lactic acid content, between the different types of whey. At pH 6.0 or higher, much of the calcium milk is retained in the cheese. The high content of calcium in the whey seems to be the main reason for variations in the physicochemical properties of whey, including its substantially lower stability, compared to that of sweet whey2. Due to the fact that the composition of demineralised whey resembles much of that of breast milk, about 65% of the total amount of low-whey mineral is used for the manufacture of infant formulas. Also, demineralised sweet whey (25–65% demineralisation) can be used in a healthy diet, being recommended for weight loss and for its curative and palliative effects in various types of degenerative chronic diseases, such as diabetes. Demineralised whey products have also been used in a range of dairy beverages2.
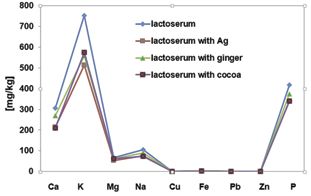
Fig. 3 Compositions of minerals identified in new dairy lactoserum beverages
The ICP-AES data reveal a calcium concentration of 305.6 mg kg–1 (0.03%) for the initial lactoserum, and of 209.7–214.7 mg kg–1 in the other three variants; these lower calcium concentrations are due most probably to the dilution of the initial lactoserum by the addition of liquid extracts. Like in the calcium case, the highest values of concentrations for the other investigated elements in mg kg–1 were found in initial lactoserum for K (751.8), Mg (67.4), Na (104.7), P (418.4), Cu (0.9), Fe (2.4), Pb (0.02) and Zn (0.6), respectively. The concentration of Ag found in Z-CS beverage is 2 mg kg–1. Regarding the concentration of the potassium in the beverage obtained from Z-CE-H, this is slightly higher than the other beverages obtained from different lactoserum (573.9 mg kg–1), probably due to the potassium concentration, known to be much higher in the cacao powder and in honey than in ginger root concentrate, where is lower than in the simple lactoserum because of its dilution by extracts adding. Similarly, the concentrations of the other minerals in all investigated beverages have close values to those of the starting lactoserum and we can assume these differences are due to dilution of the starting serum by adding the extracts.
Conclusions
The new dairy beverages based on lactoserum found on the Romanian market have similar physicochemical characteristics to sweet whey. But instead, they have a low protein and mineral content and a moderate quantity of lactose, which recommend them for curative and palliative purposes in medical fields such as obesity, diabetes mellitus type II or kidney diseases. In order to ensure their bioactive properties, more chemical and biological investigations are needed.
Acknowledgements
This research was funded by the Romanian Ministry of Education and Research, through a Research Grant (PROFI-ZONAR), PNIII-P2-2.1-BG-2016-0204 / 112 BG/2016, http://www.usamvcluj.ro/profi-zonar/; PNIII-P2-2.1-BG-2016-0335 / 28BG/2016.
References
- G. W. SMITHERS: Whey and Whey Proteins – from “Gutter-to-Gold”. Int Dairy J, 18 (7), 695 (2008).
- V. KUMAR: Module 3: Processing and Utilization of Whey. Lesson 20. Physicochemical Characteristics of Whey. Lesson 21. Manufacture of Condensed Whey Products. ICAR eCourseby AgriMoon Team, April 2016.
- K. DINKOV, T. ZLATEV: Ecological Efficiency under Membrane Milk Processing. J Environ Prot Ecol, 5 (2), 430 (2004).
- R. HASALLIU, E. BELI, J. TERPOLLARI, Xh. HAMITI: Effect of pH of Cheeses on the Growth of Staphylococcus aureus. J Environ Prot Ecol, 13 (1), 172 (2012).
- S. S. ZOELLNER, A. G. CRUZ, J. A. F. FARIA, H. M. A. BOLINI, M. R. L. MOURA, L. M. J. CARVALHO, A. S. SANŤANA: Whey Beverage with Acai Pulp as a Food Carrier of Probiotic Bacteria. Aust J Dairy Technol, 64, 165 (2009).
- K. KARABODUK, O. KARABACAK, H. KARABODUK, T. TEKINAY: Chemical Analysis and Antimicrobial Activities of the Origanum vulgare subsp. hirtum. J Environ Prot Ecol, 15 (3A), 1292 (2014).
- B. A. FOLASHADE, T. N. FAGBEMI, B. O. T. IFESAN, A. A. BADEJO: Antioxidant Properties of Cold and Hot Water Extracts of Cocoa, Hibiscus Flower Extract, and Ginger Beverage Blends. Food Res Int, 52, 490 (2013).
- M. MAJEED, L. PRAKASH: Ginger (Zingiber officinale): Product Write UP. Sabinsa Corporation – UT, 2007.
- F. A. FIORDA, G. VINÍCIUS DE MELO PEREIRA, THOMAZ-SOCCOL, A. P. MEDEIROS, S. K. RAKSHIT, C. R. SOCCOL: Development of Kefir-based Probiotic Beverages with DNA Protection and Antioxidant Activities Using Soybean Hydrolyzed Extract, Colostrum and Honey. LWT – Food Sci Technol, 68, 690 (2016).
- P. Kr. BISWAS, S. DEY: Effects and Applications of Silver Nanoparticles in Different Fields. Int J Recent Sci Res, 6 (8), 5880 (2015).
- Manual of Methods of Analysis of Foods, Milk_and_Milk_Products. FSSAI, New Delhi – 110002, India, 2016.
- M. M. BRADFORD: A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal Biochem, 72, 248 (1976).
- R. CHANDAN: Dairy-based Ingredients, CHAPTER 1: Properties of Milk and Its Components. AACCI Grain Science Library, Online Book.
- C. C. CHEN, S. T. CHEN, J. FENG HSIEH: Proteomic Analysis of Polysaccharide-Milk Protein Interactions Induced by Chitosan. Molecules, 20 (5), 7737 (2015).
- E. ROJAS, G. TORRES: Isolation and Recovery of Glycomacropeptide from Milk Whey by Means of Thermal Treatment. Food Sci Technol (Campinas), 33 (1), 14 (2013).
- www.angelfire.com/zine2/mr_x/WheyProteinByMrX.pdf.
- http://www.dairyforall.com/whey.php.
- B. DEC, W. CHOJNOWSKIȘ: Characteristics of Acid Whey Powder Partially Demineralised by Nanofiltration. Pol J Food Nutr Sci, 15 (56), SI 1, 87 (2006).
- E. SUÁREZ, A. LOBO, S. ÁLVAREZ, F. A. RIERA, R. ÁLVAREZ: Partial Demineralisation of Whey and Milk Ultrafiltration Permeate by Nanofiltration at Pilot-Plant Scale. Desalination, 198, 274 (2006).